Abstract
Introduction Tyrosine kinase inhibitor (TKI) therapy is the gold standard for patients (pts) with chronic myeloid leukemia in the chronic phase (CML-CP); however, there are few studies that demonstrate real-world long-term survival outcomes for individual TKIs (Osman Blood Rev 2021). In the SIMPLICITY (NCT01244750) study, patients with CML-CP who remained on their first-line (1L) TKI were significantly more likely to achieve clinical response compared with patients who switched to a second-line TKI; 3-year survival rates were similar (Gambacorti-Passerini Eur J Haematol 2021). This analysis of SIMPLICITY was undertaken to evaluate the impact of 1L TKIs on real-world 5-year survival among patients with CML-CP who were treated with first-generation (1G) TKI imatinib (IM) or second-generation (2G) TKIs dasatinib (DAS) or nilotinib (NIL).
Methods The observational SIMPLICITY study followed pts with CML-CP from routine clinical practice who received 1L IM, DAS, or NIL in the USA and Europe. US pts were monitored at 6-month intervals for ≤ 5 years and stratified by index TKI(s); this cohort is the focus of this analysis. Descriptive statistics and multivariate analysis are presented for prospective US pts who did not switch their 1L TKI to minimize the confounding effect from subsequent lines of therapy. The Kaplan-Meier survival estimate was calculated for overall survival (OS) at 5 years. The Cox survival analysis of OS was adjusted for age, sex, comorbidities, and 1L TKI.
Results Of 997 US pts, 426 pts did not switch from their initial 1L TKI, 308 changed TKI treatment, and 284 were not followed on study for 5 full years. Among patients who did not switch (data cutoff January 28, 2020), 113 (26.5%) received IM and 313 (73.5%) received DAS/NIL (161 [37.8%] received DAS and 152 [35.7%] received NIL) Overall, 53.8% of pts were male (55.8% of pts treated with IM and 53.0% of pts treated with DAS/NIL). The mean age at diagnosis was 54.4 (standard deviation [SD], 14.5; range, 19.1-90.7) years (57.8 [SD, 14.7] years in pts treated with IM and 53.1 [SD, 14.3] years in pts treated with DAS/NIL). Most pts (65.7%) were of White race and non-Hispanic ethnicity (62.8% of pts treated with IM and 66.8% of pts treated with DAS/NIL). Overall, 43.4% of pts were treated at academic centers (35.4% of pts treated with IM and 46.3% pts treated with DAS/NIL). Among pts whose Eastern Cooperative Oncology Group (ECOG) performance status was reported (n = 234), 62.8% had a score of 0 (51.7% of pts treated with IM and 66.7% pts treated with DAS/NIL). The mean number of comorbid conditions was 3.3 (SD, 2.8; range, 0.0-15.0) overall (3.5 [SD, 2.7; range, 0.0-12.0] in pts treated with IM and 3.3 [SD, 2.8; range, 0.0-15.0] pts treated with DAS/NIL).
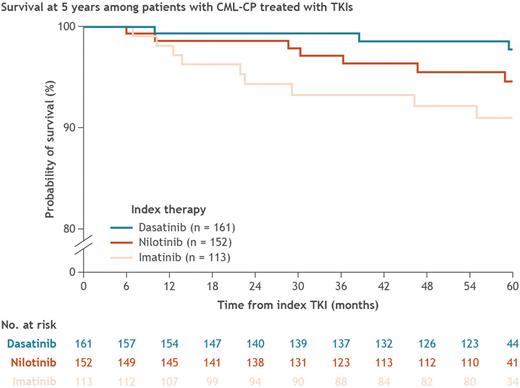
The 5-year crude OS rate was 94.9% (standard error [SE], 1.2) overall (91.0% [SE, 2.9] in pts treated with IM and 96.3% [SE, 1.2] in pts treated with DAS/NIL). Notably, the difference in survival rates appeared to increase across successive time points (Figure). Nineteen pts died during the study (9 pts treated with IM and 10 pts treated with DAS/NIL). Of these, 11 (57.9%) pts had a reported known reason for death, with death related to CML occurring in 0 pts in the IM cohort and 1 (5.3%) pt in the DAS/NIL cohort; most deaths were not related to CML (10/19 [52.6%] pts overall (5/9 [55.6%] pts treated with IM and 5/10 pts treated with DAS/NIL [50.0%]). Cox proportional hazard model analysis showed that, compared with pts treated with IM, pts treated with DAS had a 59% reduction in risk of death during the observation period (hazard ratio [HR], 0.41; 95% confidence interval [CI], 0.10-1.63; P = 0.21), and pts treated with NIL had a 16% reduction in risk of death (HR, 0.84; 95% CI, 0.31-2.33; P = 0.74).
Conclusions Among pts with CML-CP who did not switch 1L treatment, the SIMPLICITY study showed high 5-year OS rates regardless of initial treatment. This study revealed that pts treated with IM were predominantly treated at non-academic centers, were generally older, were less likely to be of White race and non-Hispanic ethnicity, and had higher ECOG scores compared with pts treated with DAS/NIL. The survival results in the figure are descriptive, reflecting the unequal patient cohort composition. Future studies are needed to evaluate long-term survival in larger cohorts of pts treated with IM and other TKIs.
Study support: This study was funded by Bristol Myers Squibb.
Disclosures
Mauro:AbbVie, Bristol Myers Squibb, Novartis, Pfizer, Takeda: Consultancy, Honoraria, Other: Travel, accommodation, expenses , Research Funding; Sun Pharma/SPARC: Research Funding. Paquette:Novartis: Consultancy. Goldberg:COTA Healthcare: Current equity holder in private company, Current holder of stock options in a privately-held company; MDS Foundation: Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy. Tang:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. DeGutis:Bristol Myers Squibb: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. McBride:Bristol Myers Squibb: Current Employment. Parsons:ICON Clinical Research: Current Employment. Montelongo:ICON plc: Current Employment. Cortes:Novartis: Consultancy, Honoraria, Research Funding; Gilead: Consultancy; Abbvie: Consultancy, Research Funding; Biopath Holdings Inc: Consultancy, Current equity holder in private company; Sun Pharma: Consultancy, Research Funding; Takeda: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria, Research Funding; Forma Therapeutic: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal